3.1 Special Requirement for Production
Non-final sterilizing aseptic packing injection means the products are produced by aseptic process without heating sterilizing.
3.1.1 The injections, which require aseptic packing, are heat-hating products. Final heating sterilizing could not be adopted. Aseptic operation during producing process should be enforced, and any contamination should be prevented.
3.1.2 For this kind of products, the damp-absorbing characteristic is strong. During production, special attention should be paid to the relative humidity of aseptic room, water in the rubber plug and bottle, tools' dryness and packing tightness.
3.1.3 For ensuring aseptic character of the products, the air purity of sanitary room and air-condition cleaning system's running should be supervised strictly all the time. The aseptic operations and non-aseptic operations should be divided strictly. Anything should be sterilized before enter the aseptic room. The operators should comply with the standards for aseptic industry (SOP).
3.1.4 Any equipment and processes, which may affect the quality of products, should be validated and supervised. The valid period of sterilized packing equipment and other relative article, which contact the products directly, should be limited and regulated. ˇŁ
3.2 Flow sheet of aseptic packing injection
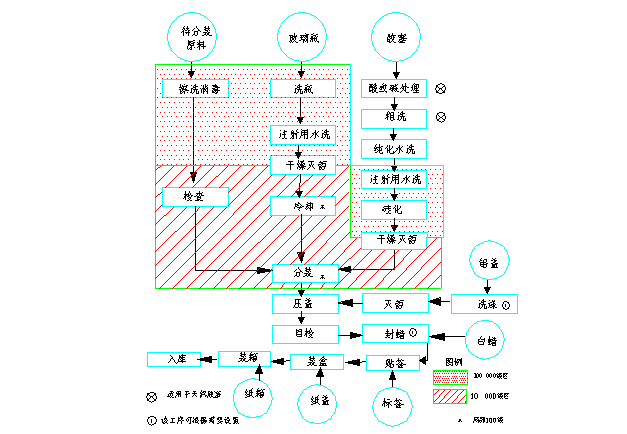
|